|
The biological effects of SARS-CoV-2 are governed by the interaction of the homotrimeric spike (S) glycoprotein with cognate receptors on human cells. Interestingly, a reduction in the vascular coverage by PCs was documented in the heart and lungs of human patients with COVID-19, in the absence of capillary rarefaction, suggesting that SARS-CoV-2 may affect the microvasculature by specifically targeting PCs. Cardiac myocytes and fibroblasts express the main entry receptor angiotensin-converting enzyme 2 (ACE2), but pericytes (PCs), mural cells that support the maintenance and repair of microvasculature throughout the myocardium, appear particularly susceptible because they reportedly express the highest levels of ACE2 in the heart. While nasal and pulmonary epithelial cells are the primary target for infection, after viral replication and circulation, many other cells in distant organs, including heart resident cells, become exposed and potentially infected. The link between the two conditions is not completely understood, especially in the light of the controversy about the capacity of the coronavirus to infect the vascular endothelium. Moreover, people with pre-existing cardiovascular diseases are more likely to die of COVID-19. Microvascular complications are frequent and harmful in patients with coronavirus disease-2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), with up to 11% of those hospitalised in intensive care units having myocardial ischaemia or infarction. This mechanism may have clinical and therapeutic implications. In conclusion, our findings suggest that the S protein may prompt PC dysfunction, potentially contributing to microvascular injury. Immunoreactive S protein was detected in the peripheral blood of infected patients. The neutralisation of CD147, either using a blocking antibody or mRNA silencing, reduced ERK1/2 activation, and rescued PC function in the presence of the S protein. 
Next, adopting a blocking strategy against the S protein receptors angiotensin-converting enzyme 2 (ACE2) and CD147, we discovered that the S protein stimulates the phosphorylation/activation of the extracellular signal-regulated kinase 1/2 (ERK1/2) through the CD147 receptor, but not ACE2, in PCs. Exposure to the recombinant S protein alone elicited signalling and functional alterations, including: (1) increased migration, (2) reduced ability to support endothelial cell (EC) network formation on Matrigel, (3) secretion of pro-inflammatory molecules typically involved in the cytokine storm, and (4) production of pro-apoptotic factors causing EC death. Here we newly show that the in vitro exposure of primary human cardiac PCs to the SARS-CoV-2 wildtype strain or the α and δ variants caused rare infection events. We tested this hypothesis in pericytes (PCs), which are reportedly reduced in the heart of patients with severe coronavirus disease-2019 (COVID-19). Additionally, the SARS-CoV-2 Spike (S) protein may act as a ligand to induce non-infective cellular stress. 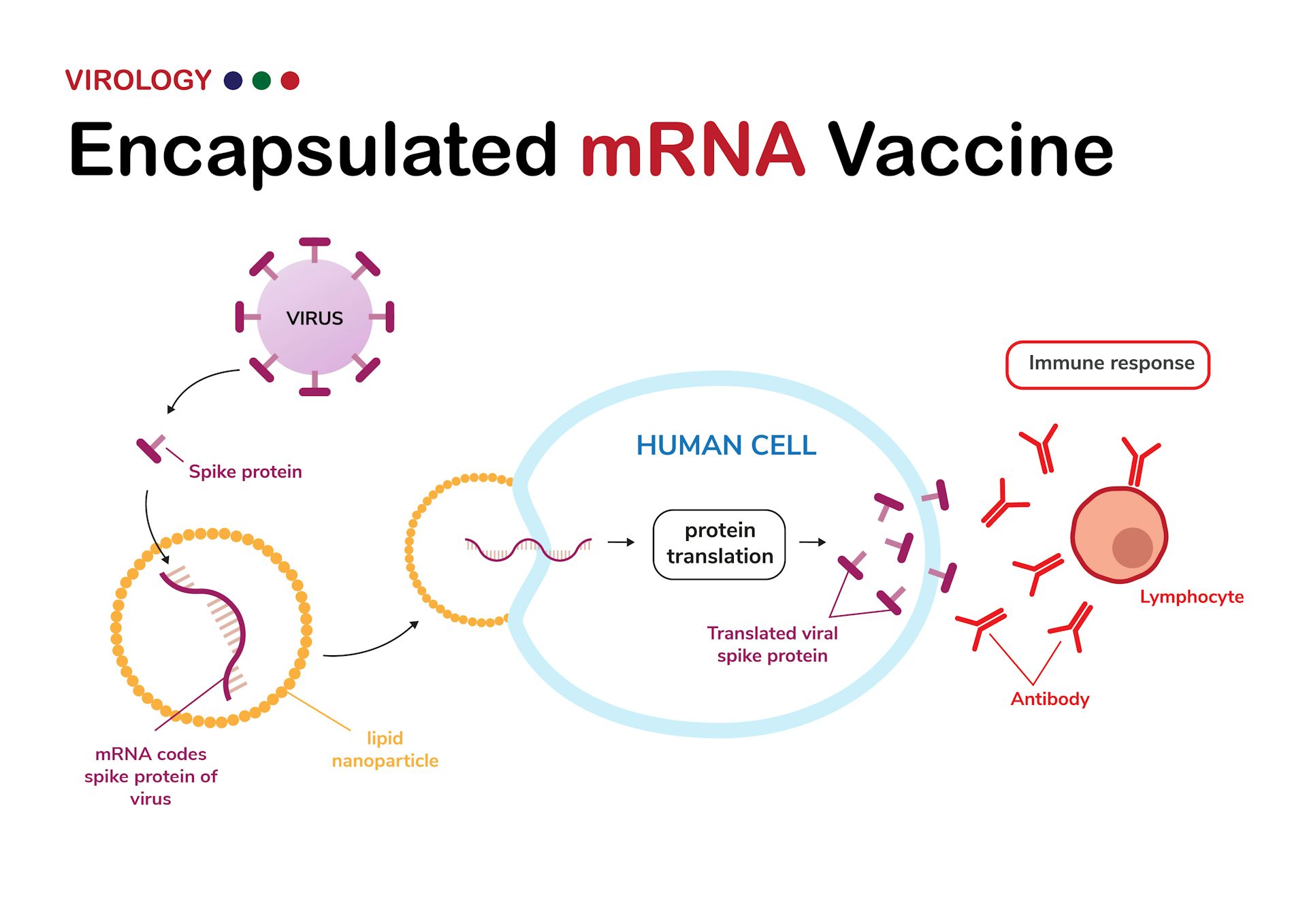
The capacity of SARS-CoV-2 to infect vascular cells is still debated. The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes a broad range of clinical responses including prominent microvascular damage.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
